Soil scientists and agronomists who criticise the Albrecht system of soil mineral balancing speak of the ‘calcium/magnesium ratio myth’ – and they’re right. There is a myth. But it exists only in their minds and is due to their misunderstanding the ideas of Professor William Albrecht. Neal Kinsey, author of Hands-on Agronomy, was Prof Albrecht’s last student before he retired. Since then, Neal has worked with farmers all over the world to successfully apply the Albrecht principles.
I’ve attended three of Neal’s advanced courses, one in Australia, one in the US and one in South Africa. He has also visited SA seven times in the past 12 years to conduct courses and give talks at biological farming forums. The point I want to make is that I’ve never heard him refer to the ‘calcium:magnesium ratio’.
Ideal amount
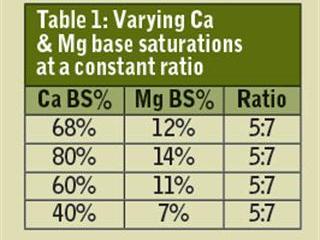
Albrecht specified the ideal amount of calcium and magnesium required to create a healthy soil. This amount is best expressed in terms of the percentage of these positively-charged minerals which attach themselves to the negative charges on soil colloids. This is the base saturation (BS) level. For loam and clay soils, Albrecht recommends that 68% of the total negative charges should be saturated with calcium and 12% with magnesium. Note that 68% + 12% = 80%.
The remaining 20% should be saturated with potassium, sodium, hydrogen (required for acidity) as well as copper, zinc, manganese, iron, aluminium and other positively charge trace elements. For sandy soils, Prof Albrecht prescribed a range of calcium and magnesium base saturations, depending on how sandy the soil is.
I won’t delve into this now as I need to make the point that using ratios to analyse or predict soil fertility can be horrendously misleading. Now, as noted, the ideal calcium and magnesium BS levels for loam and clay soils are 68% and 12% respectively. As can be seen in Table 1 the ratio of Ca to Mg works out to 5:7 in this specific case, which is ideal.

Compromised
But what happens where dolomitic lime has been over-applied to such an extent that the Ca and Mg BS levels increase to 80% and 14% respectively? The ratio still works out to 5:7, but the soil’s productive capacity has been seriously compromised – and crop yields will drop for a range of reasons. The most obvious of these is that calcium and magnesium will now saturate 94% of the negative charges on the soil colloid, leaving only 6% for all the other important plant nutrients. Said in another way, an excess of calcium will limit plant uptake of all other essential minerals.
Major shortage
What happens when there’s a major shortage of calcium and magnesium – when the base saturations are, say 40% and 7%? Once again, the ratio is 5:7, but this soil will have an extremely low level of productivity. Apart from having far too little calcium, which will restrict plant productivity, the major problem is the very low magnesium level. Magnesium is the heart of chlorophyll, where solar energy is bound out of carbon dioxide, air and water – certainly, the cheapest and most productive power station in the world!
Albrecht revealed that, as soon as Mg BS% drops below 11%, plants start running out of energy; at 7%, the leaf ‘power station’ can’t meet plant demands. It should now be obvious that it’s sheer folly to either predict or analyse crop yields based on a nebulous ‘calcium:magnesium ratio’, never mind the absurdity of using it to discredit the Albrecht system.
John Fair is a leading expert on pastures. He heads up Fair’s Biofarm Assist, and can be contacted on 058 622 3585 or [email protected].










